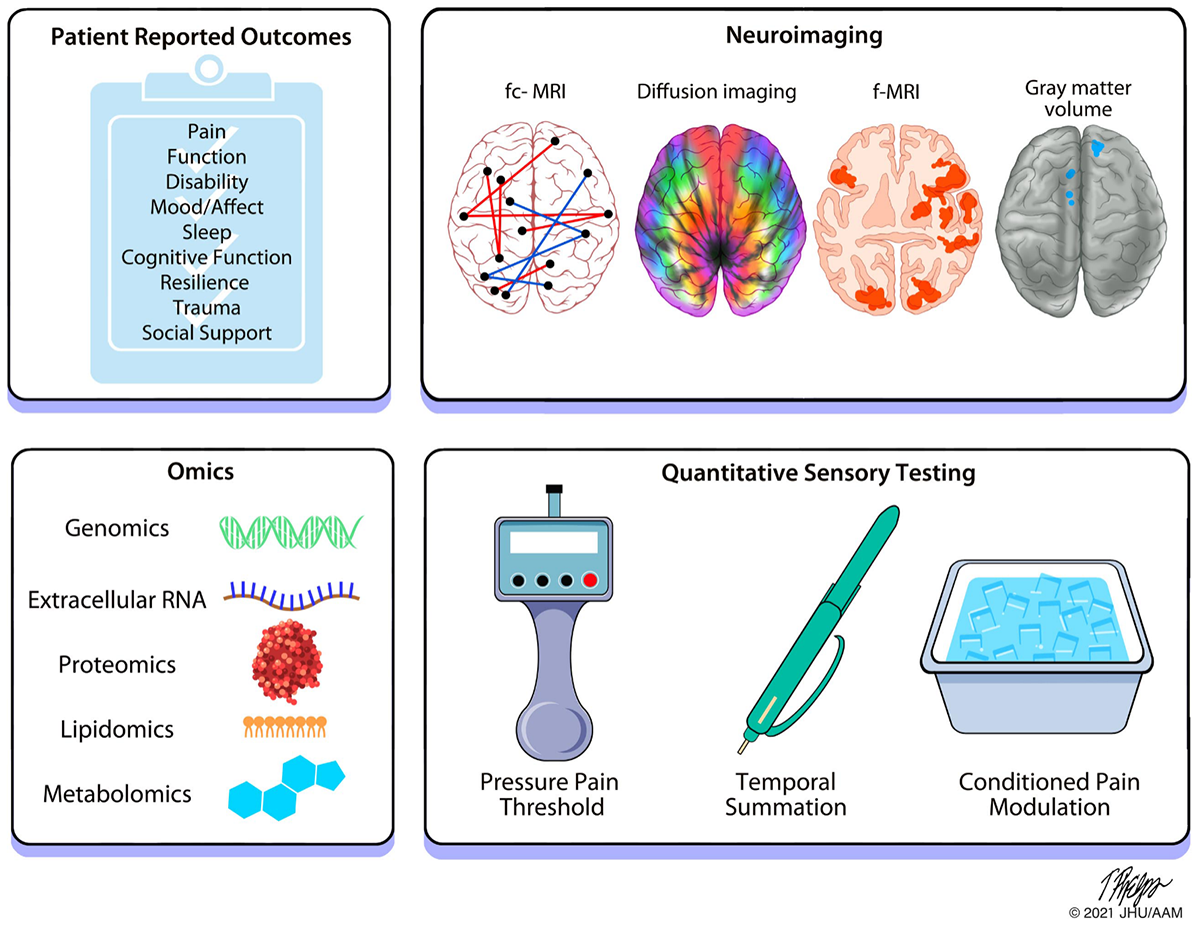
Biomarkers

A core aim of the A2CPS project is to identify biomarkers of chronic pain. Biomarkers are objectively measurable signs of a medical condition, such as high blood glucose in diabetes. No such biomarkers yet exist for pain, but they could help doctors and researchers identify patients who are at high risk of developing chronic pain, or track signs of improvement in response to a treatment. A2CPS applies a team-science, consortium-based approach to validate and discover biomarkers and biosignatures for the transition to chronic pain using cutting-edge techniques across multiple domains. Although the FDA defines biomarkers as "molecular, histologic, radiographic or physiologic characteristics that provide an indication of biological or pathogenic processes," the A2CPS consortium considers markers across biologic, psychosocial and clinical domains for chronic post-surgical pain. The study will assess 38 putative biomarkers, which were carefully chosen based on existing evidence, scalability to A2CPS, and clinical utility. A2CPS hopes to find signatures, made up of multiple biomarkers, that emerge in patients with chronic post-surgical pain (CPSP) in hopes that they could one day help identify therapeutic targets, or help researchers predict who might develop post-surgical pain in the future. A2CPS biomarker signatures may be made up of multi-omics data, brain imaging, and psychosocial and sensory measures. To learn more about biomarkers, check out the BEST (Biomarkers, EndpointS and other Tools) resource from the FDA and the NIH.